



To receive important alerts and updates from Gold Coast Primary Health Network, please submit the form below.
"*" indicates required fields

To receive important alerts and updates from Gold Coast Primary Health Network, please submit the form below.
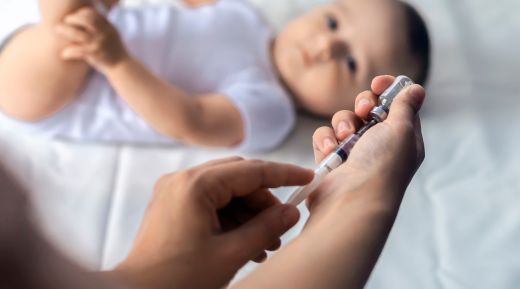
The Department of Health, Disability and Ageing (DHDA) is updating the childhood pneumococcal vaccination schedule on the National Immunisation Program (NIP), based on advice from the Australian Technical Advisory Group on Immunisation (ATAGI). Starting September 1, 2025, a new vaccine, Prevenar 20, will replace both Prevenar 13 and Pneumovax 23 on the childhood schedule.
This change aims to simplify the vaccination schedule, reduce the total number of required doses for some children, and provide broader protection against more strains of the disease. Queensland has been distributing the new vaccine to providers since Monday, August 18, to ensure all clinics have stock for the September 1 commencement date.
Here is a breakdown of the new schedule effective September 1, 2025:
1. Non-First Nations children without medical risk conditions: The dose frequency remains a 2+1 schedule (two primary doses plus one booster).
2. First Nations children: The dose frequency remains a 3+1 schedule (three primary doses plus one booster). This is unchanged in QLD, but other states are now aligned for National consistency.
3. Children with medical risk conditions aged 2-12 months: Children in this group will continue to follow a 3+1 schedule, three primary doses and one booster.
4. Children and adolescents aged over 12 months to under 18 years with specified medical risk conditions: This group should now receive a single dose of Prevenar 20 at diagnosis, replacing the previous schedule of Prevenar 13 followed by two doses of Pneumovax 23.
5. Removal of Pneumovax 23 from the childhood schedule: Pneumovax 23 for children is being discontinued. For eligible children (medically at risk or First Nation) that have completed a Prevenar 13 course and are due for Pneumovax 23, they should receive a single dose of Prevenar 20 instead.
Summary
Effective 1 September 2025, the NIP-funded childhood pneumococcal vaccine schedule will be simplified and standardised nationwide with the introduction of Prevenar 20, replacing both Prevenar 13 and Pneumovax 23. Prevenar 20 offers broader protection against 20 strains and will be administered at 2, 4, and 12 months for non-First Nations children without risk conditions, and at 2, 4, 6, and 12 months for First Nations children and those with medical risk factors. Children who have already started their schedule with Prevenar 13 will transition to Prevenar 20 for remaining doses, and those previously due for Pneumovax 23 will now receive a single dose of Prevenar 20.
Resources:
Department of Health, Disability and Ageing:
National Centre of Immunisation Research and Surveillance (NCIRS):
Queensland Health
So we can provide you with the most accurate information,
please tell us a little more about yourself

